A Mechanistic Investigation of the Kinetic Resolution of Secondary Aromatic Alcohols Using a Ferrocene‐Based Planar Chiral 4‐(Dimethylamino)pyridine Catalyst - Mesas‐Sánchez - 2015 - Chemistry – A European Journal - Wiley Online Library
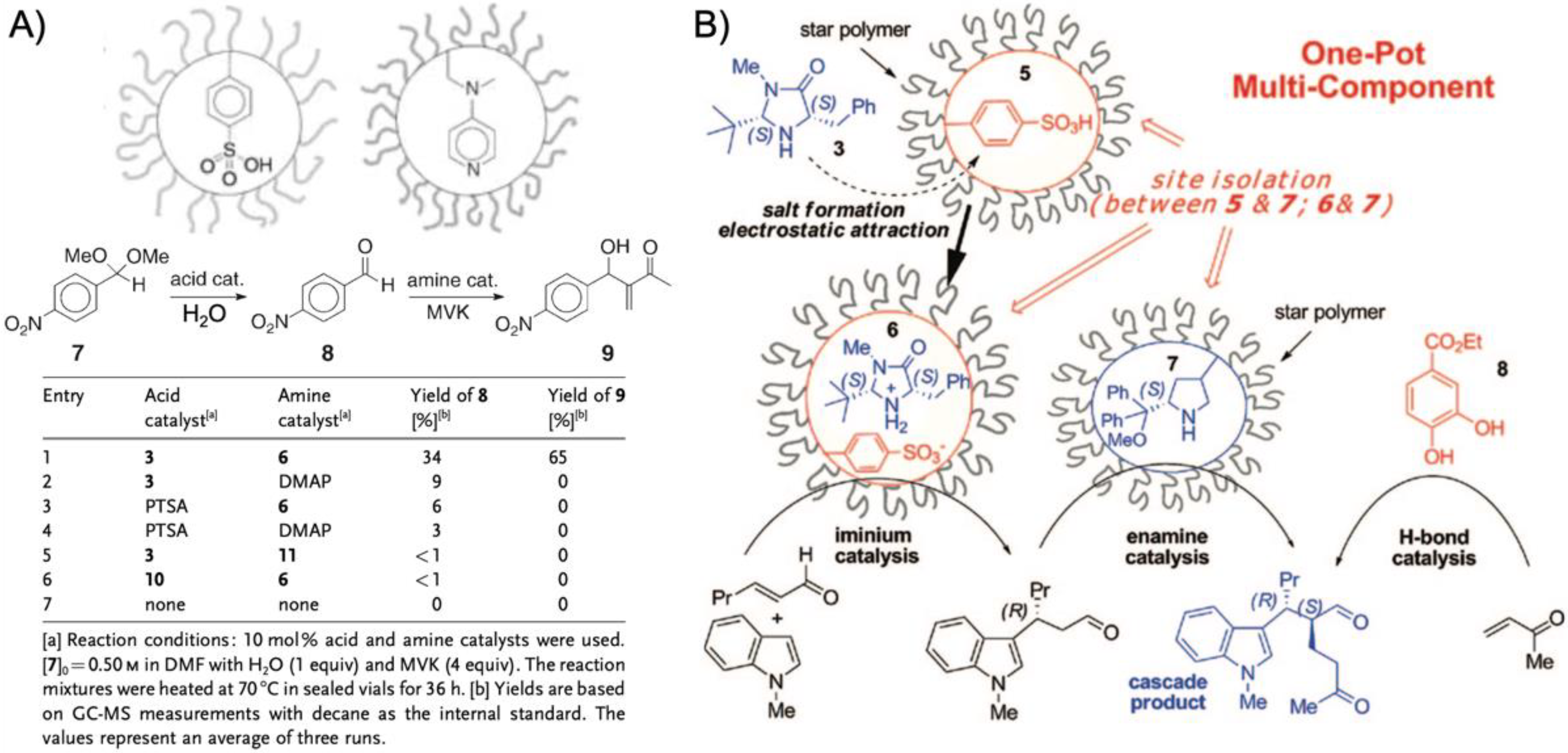
Polymers | Free Full-Text | Recent Advances in the Synthesis and Application of Polymer Compartments for Catalysis | HTML
Chapter 1 Nucleophilic Catalysts and Organocatalyzed Zwitterionic Ring-opening Polymerization of Heterocyclic Monomers (RSC Publishing) DOI:10.1039/9781788015738-00001

Catalysts | Free Full-Text | Organocatalysis: Fundamentals and Comparisons to Metal and Enzyme Catalysis | HTML
![Scheme 1. Proposed Mechanism for [Cu(MeCN) 4 ]PF 6 / DBED/DMAP Catalyst... | Download Scientific Diagram Scheme 1. Proposed Mechanism for [Cu(MeCN) 4 ]PF 6 / DBED/DMAP Catalyst... | Download Scientific Diagram](https://www.researchgate.net/publication/313971430/figure/fig3/AS:465341852721154@1487957551725/Scheme-1-Proposed-Mechanism-for-CuMeCN-4-PF-6-DBED-DMAP-Catalyst-System.png)
Scheme 1. Proposed Mechanism for [Cu(MeCN) 4 ]PF 6 / DBED/DMAP Catalyst... | Download Scientific Diagram

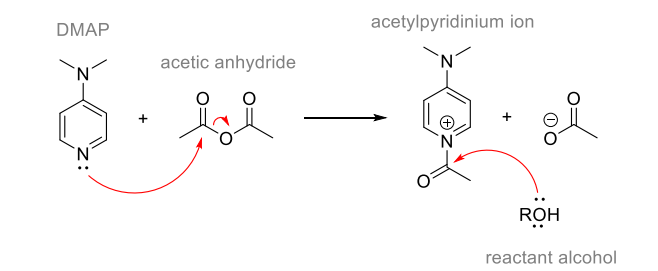
Nucleophilic Catalysis by 4‐(Dialkylamino)pyridines Revisited—The Search for Optimal Reactivity and Selectivity - Spivey - 2004 - Angewandte Chemie International Edition - Wiley Online Library
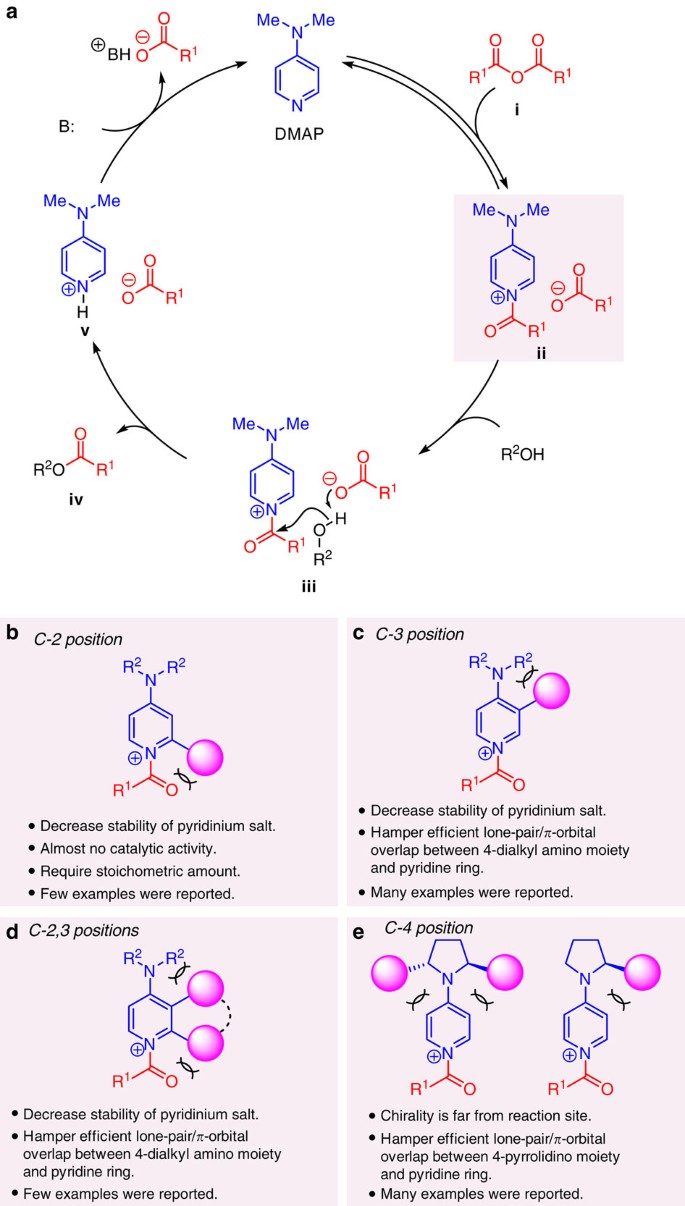
Enantioselective acyl transfer catalysis by a combination of common catalytic motifs and electrostatic interactions | Nature Communications
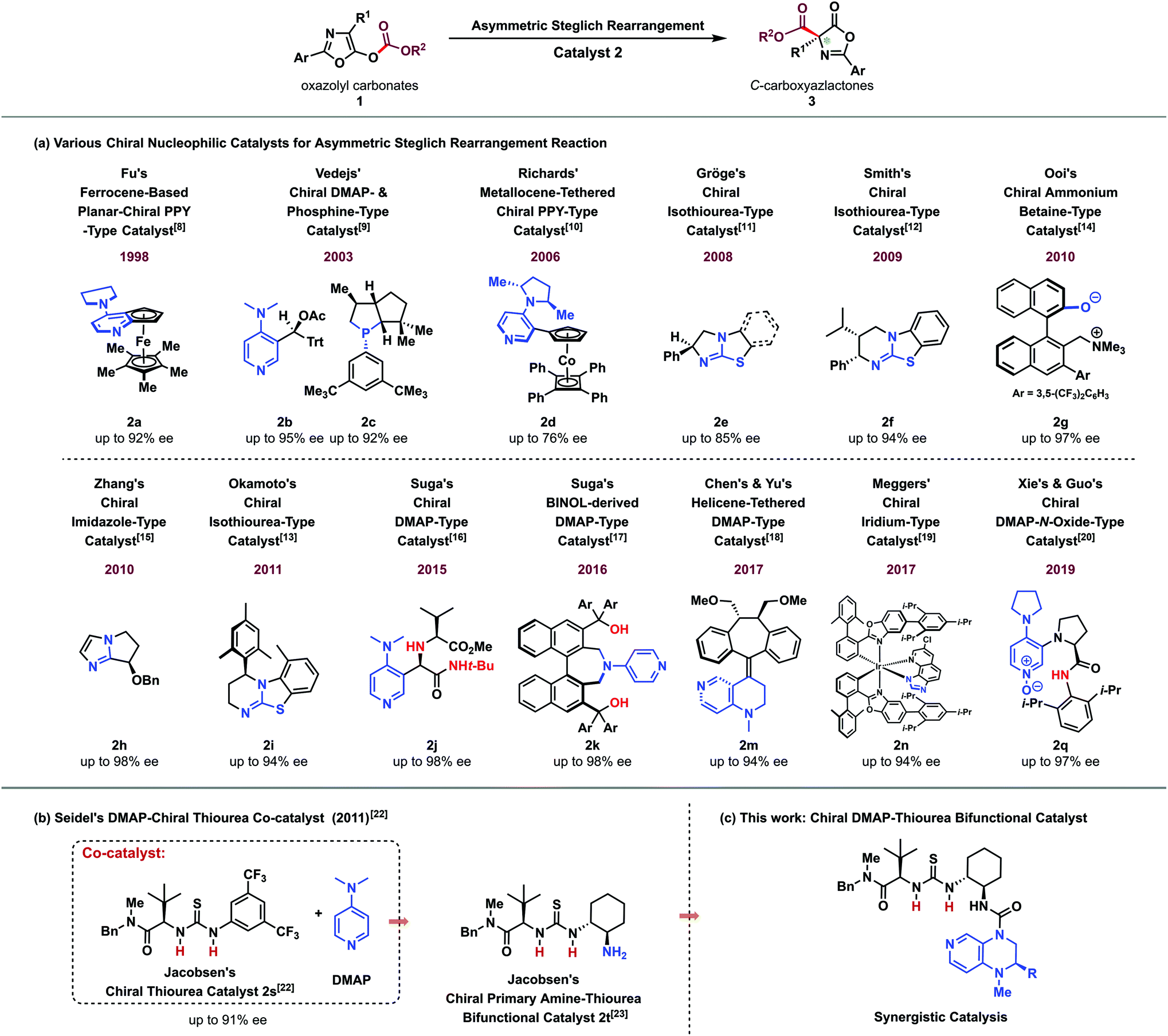
A novel chiral DMAP–thiourea bifunctional catalyst catalyzed enantioselective Steglich and Black rearrangement reactions - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/C9QO00691E

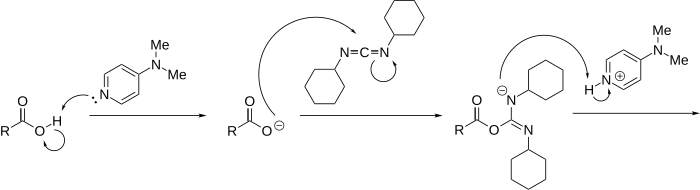
Scheme IV.5: Mechanism of activation with DCC using DMAP as a catalyst. | Download Scientific Diagram
Scheme 1 Mechanism of the formation of E-CNCs with DMAP as a catalyst. | Download Scientific Diagram

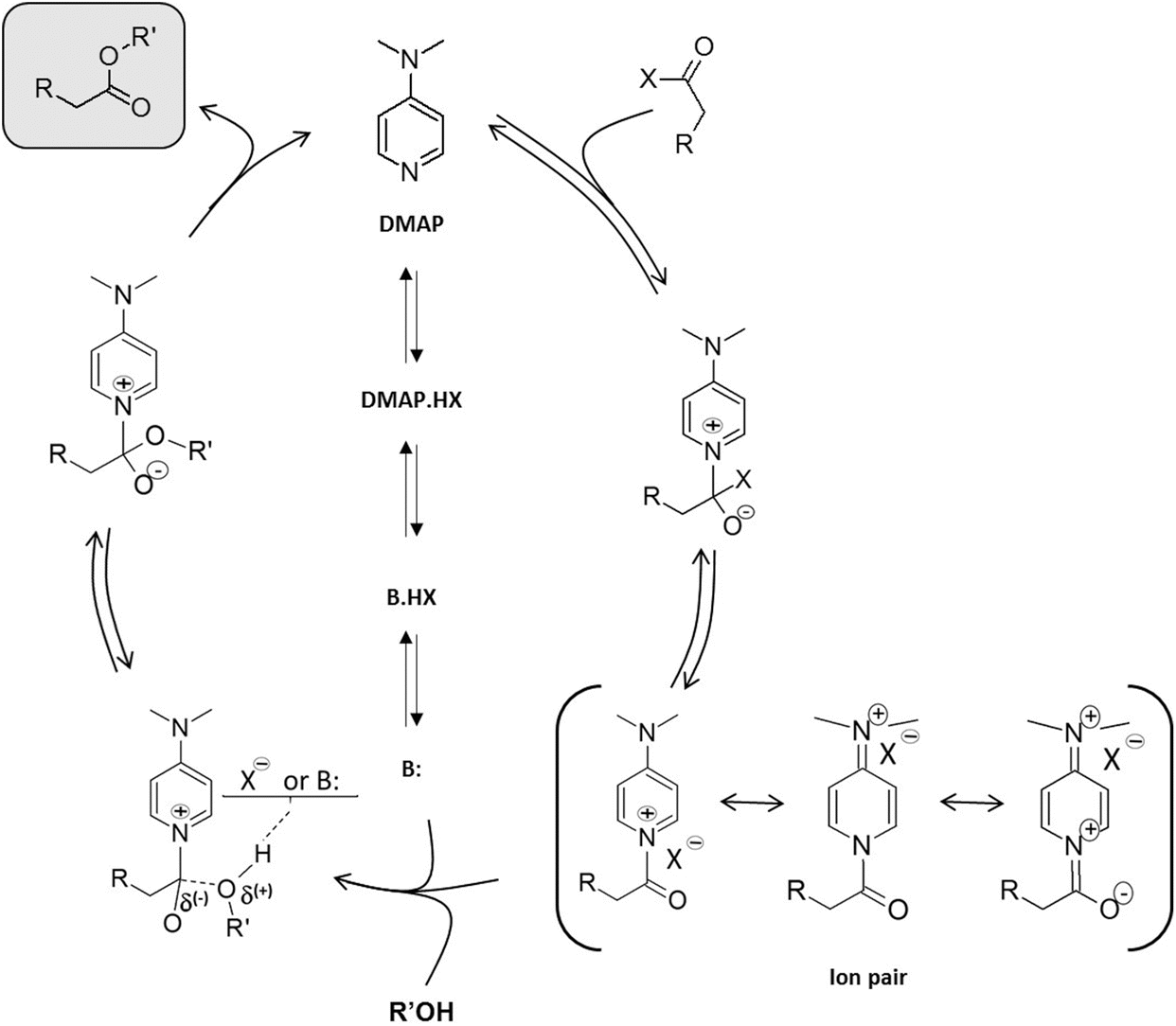
Steric Effects in the Uncatalyzed and DMAP‐Catalyzed Acylation of Alcohols—Quantifying the Window of Opportunity in Kinetic Resolution Experiments - Fischer - 2006 - Chemistry – A European Journal - Wiley Online Library


![Scheme 2 The proposed mechanism for the DMAP-catalyzed [2 + 4]... | Download Scientific Diagram Scheme 2 The proposed mechanism for the DMAP-catalyzed [2 + 4]... | Download Scientific Diagram](https://www.researchgate.net/profile/Shiwen-Du/publication/307905424/figure/fig1/AS:481870543626240@1491898298068/Scheme-2-The-proposed-mechanism-for-the-DMAP-catalyzed-2-4-cycloaddition-reaction-in.png)